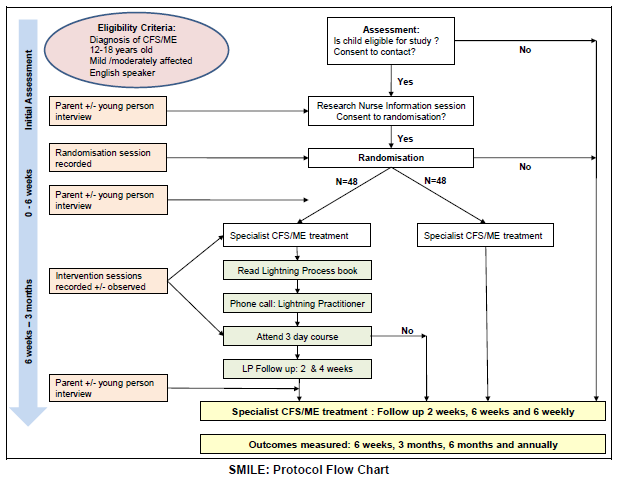
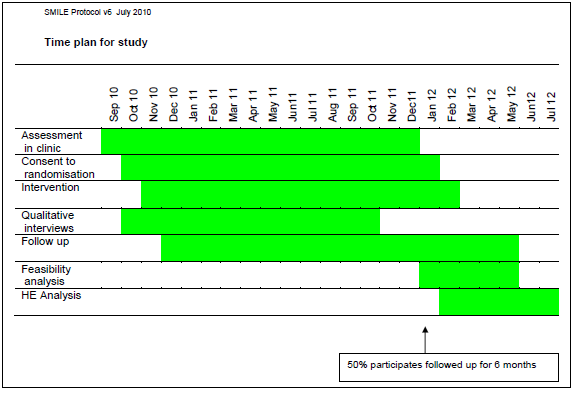
SMILE – Specialist Medical Intervention and Lightning Evaluation documents (Lightning Process pilot study – children [now aged 12 to 18] with CFS and ME)
Shortlink: http://wp.me/p5foE-37x
See also previous ME agenda post:
“Unethical” Lightning Process pilot study in children receives ethics approval
Update: Key documents
3] SMILE Research Protocol
Open here: smprotv6final
29] Research Ethics Application Form
Open here: recfrmrfs
University of Bristol website
http://www.bristol.ac.uk/ccah/research/childrencomplexhealthneeds/chronic-fatigue/smile.html
Source: Protocol document
SMILE – Specialist Medical Intervention and Lightning Evaluation
What is SMILE?
SMILE is a feasibility study to see whether it is possible to recruit young people into a study to compare specialist medical treatment with specialist medical treatment plus the Lightning Process for young people with chronic fatigue syndrome or ME (CFS/ME).
The study will also look at how we should measure outcomes and the health economic impact on the families of young people with CFS/ME.
Young people will be observed completing the questionnaires that we use to look at how unwell they are before they see us and what happens to them after an intervention. We will also talk to young people and their parents to understand what they think about the questionnaires and to determine the most acceptable and sensitive ones to use. This study is the first to work out which questionnaires we should be using to understand outcome in paediatric CFS/ME.
Frequently Asked Questions
Why is research in children needed?
Over 250 children a year already attend Lightning Process training. It is important that people know whether it is safe and effective or not. We need high quality research to answer these questions. If SMILE can recruit enough people to participate in the study then further research could look at whether it is helpful or not.
Should research be done in children before adults?
Children have the right to research particularly in illnesses which are different to adults. CFS/ME in children has a different outcome to adults and the treatment is different therefore research in adults cannot be extrapolated to children.
How will the safety of those involved in SMILE be monitored?
The safety and wellbeing of people involved in any research project, not just the SMILE project, is of the utmost importance. There is an Independent Advisory Group to oversee, and monitor this research. All participants will be carefully monitored and regularly reviewed in the specialist CFS/ME service. Young people taking part can opt out of the trial at any point.
How can we take part in the study?
Young people are eligible if they are between 12 and 18 years of age, have CFS/ME and are from the region covered by the Bath/Bristol specialist CFS/ME service. Young people are recruited at assessment so you will not be eligible if you have already been seen by the service.
What ethical review has SMILE received?
The study has been scrutinised by the South West 2 Research Ethics Committee whose role it is to ensure that research is safe and ethically sound. The ethics committee have looked in detail at the study design, and all associated documentation and suggested improvements to the readability and accessibility of the patient information leaflets and consent forms which have been adopted.
The SMILE study is compliant with Good Clinical Practice Guidelines, Research Governance Framework, Medical Research Council guidelines, Royal College of Paediatrics and Child Health guidelines for the conduct of trials and has been approved by an ethics committee.
Further information about this research project can be found in the following documents:
Smile Study Documents
[Ed: I have numbered these documents for ease of reference – they are not numbered on the University of Bristol website.]
Note: some of the documents on this page are in PDF format. In order to view a PDF you will need Adobe Acrobat Reader
1] SMILE Information sheet for teenagers August 2010 [pdf (150kb)]
Open here: infoshtteensv4aug10
2] SMILE Information sheet for parents September 2010 [pdf (147kb)]
Open here: infoshtprntsv7sept10
3] SMILE Protocol Final July 2010 [pdf (170kb)]
Open here: smprotv6final
4] SMILE Under 16 assent to contact July 2010 [pdf (109kb)
Open here: u16asscv4july10
5] SMILE 16 to 18 consent to contact July 2010 [pdf (110kb)]
Open here: 16to18confinaljuly10
6] SMILE Parental consent to contact 10 May 2010 [pdf (111kb)]
Open here: parconsv310may10
7] SMILE Under 16 assent to study July 2010 [pdf (112kb)]
Open here: u16asscv4july10
8] SMILE 16 to 18 consent to study July 2010 [pdf (110kb)]
Open here: 16to18constjuly10final
9] SMILE Parental consent to study 10 May 2010 [pdf (113kb)]
Open here: parconssv310may10
10] SMILE teenager consent/assent to teenager interview August 2010 [pdf (110kb)]
Open here: tcontinvv5aug10
11] SMILE Parental consent to child interview 10 May 2010 [pdf (111kb)]
Open here: parconcinv10may10
12] SMILE Parental consent to parental interview 10 May 2010 [pdf (109kb)]
Open here: parconinvv310may10
13] SMILE Consent to record intervention for participants, parents and those delivering interventions July 2010 [pdf (110kb)]
Open here: conrecintjuly10final
14] SMILE Lightning process assessment form July 2010 [pdf (159kb)]
Open here: lipcassfrmv2july10
15] SMILE letter to GP 10 May 2010 [pdf (49kb)]
Open here: letgpv110may10
16] SMILE WPAI [pdf (135kb)]
Open here: wpai
17] SMILE Health resource use questionnaire 10 May 2010 [pdf (232kb)]
Open here: healthresuseq
18] SMILE SF-36 [pdf (165kb)]
Open here: smilesf36
19] SMILE Interview topic guide 10 May 10 [pdf (178kb)]
Open here: topgdev210may10
Correspondence with Ethics Documents
20] Initial covering letter to NREC 20th May 2010 [pdf (75kb)]
Open here: covlet20may10
21] NREC Letter 14th June 2010 [pdf (108kb)]
Open here: let14jun
22] NREC Letter 19th July 2010 [pdf (272kb)]
Open here: let19july
23] Covering letter in reply to NREC 28th July 2010 [pdf (159kb)]
Open here: letrep28july
24] NREC Letter 13th August 2010 [pdf (72kb)]
Open here: let13aug
25] Letter re meeting notes in reply to NREC 19th August 2010 [pdf (45kb)]
Open here: letmetn19aug
26] Second covering letter reply to NREC 20th August 2010 [pdf (109kb)]
Open here: seclet20aug10
27] Letter in reply to NREC 13th September 2010 [pdf ( 80kb)]
Open here: let13sep
28] NREC Approval letter 14th September 2010 [pdf (213kb)]
Open here: applet14sep10
29] REC Form [pdf (353kb)]
Open here: recfrmrfs